EU regulation
EU regulation
-
EU Traditional Herbal Registration Scheme Opens Market For Qualified TCM Products
Practical, data-backed guidance on leveraging the EU Traditional Herbal Registration (THR) scheme for qualified TCM products — including approval stats, compliance tables, and actionable next steps.
2026-03-12
-
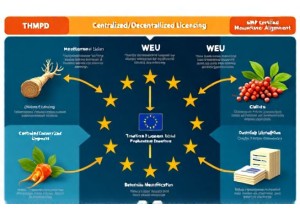
European Union Regulatory Pathways for Registered Chinese Herbal Products
Practical, data-backed guide to EU registration pathways for Chinese herbal products—including THMP, WEU, and licensing success rates, timelines, and top compliance pitfalls.
2026-03-11
-
TCM International Registration Pathways For Herbal Products In EU Markets
A practical, data-backed guide to navigating TCM international registration for herbal products in EU markets — covering THMP vs food supplement routes, costs, timelines, and real-world success tips.
2026-01-30