EMA
EMA
-
Global Regulatory Challenges in Registering Traditional Chinese Medicine as Phytopharmaceuticals
Practical analysis of regulatory hurdles for registering Traditional Chinese Medicine as phytopharmaceuticals in EU, US & Japan — with success rates, timelines, and data-driven strategies.
2026-03-22
-
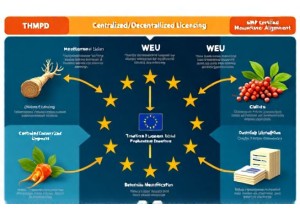
European Union Regulatory Pathways for Registered Chinese Herbal Products
Practical, data-backed guide to EU registration pathways for Chinese herbal products—including THMP, WEU, and licensing success rates, timelines, and top compliance pitfalls.
2026-03-11
-
Chinese Herbal Medicine Innovation Meets FDA EMA Regulatory Requirements
Practical insights on aligning Chinese herbal medicine innovation with FDA & EMA requirements—backed by approval data, success metrics, and actionable regulatory strategy.
2026-03-11
-
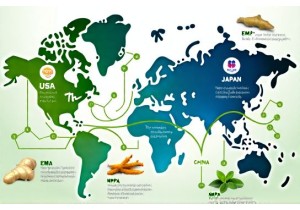
Herb Regulatory Frameworks Comparing FDA EMA and NMPA Requirements
A practical, data-driven comparison of herb regulatory frameworks across FDA (USA), EMA (EU), and NMPA (China) — with real timelines, compliance thresholds, and actionable tips for brands.
2026-02-10
-
Herb Regulatory Pathways Across US EU and Asian Market Jurisdictions
A field-tested, data-backed comparison of herb regulatory pathways across the US, EU, Japan, and China — written by a regulatory strategist with 12+ years in botanical compliance.
2026-02-09
-
Global Standards for Traditional Herbal Medicine Regulation
A field-tested, data-backed guide to navigating global traditional herbal medicine regulation — from EU THMPD to FDA DSHEA, with real compliance tables and actionable steps.
2026-01-23